Thanks to its ease of processing, ductility, and strength, steel has become an integral part of daily life and has inspired many technological innovations. However, alongside these superior properties, steel has one major weakness: due to its natural affinity for oxygen, corrosion occurs, which weakens its physical structure. The most effective way to eliminate this weakness is through metallic coating applications.
Hot-Dip Galvanizing (HDG) is a metallic coating method that has been used for approximately 150 years to protect iron and steel against corrosion. It is formed through a metallurgical reaction resulting from diffusion, where iron and steel products designed and chemically suitable for galvanizing are immersed in a molten zinc bath.
GALVANIZING PROCESS
For products whose suitability for galvanizing has been verified, the appropriate hanging method is determined.
The products are immersed in an alkaline bath to remove any oil and chemical residues that may be present on the material surface.
They are then dipped into hydrochloric acid (HCl) baths with a concentration of 18%, and kept there until any rust on the material is completely removed. Even if no visible rust exists, this acid treatment is necessary to activate the surface for galvanizing.
After the acid bath, the products are rinsed in a water bath to clean the surface.
The rinsed products are immersed in a flux bath consisting of 30% NH₄Cl–ZnCl₂ at a temperature of 50–70°C, in order to ensure proper adhesion of molten zinc to the product surface.
After flux coating, the products are dipped into a molten zinc bath at 445–465°C for galvanizing.
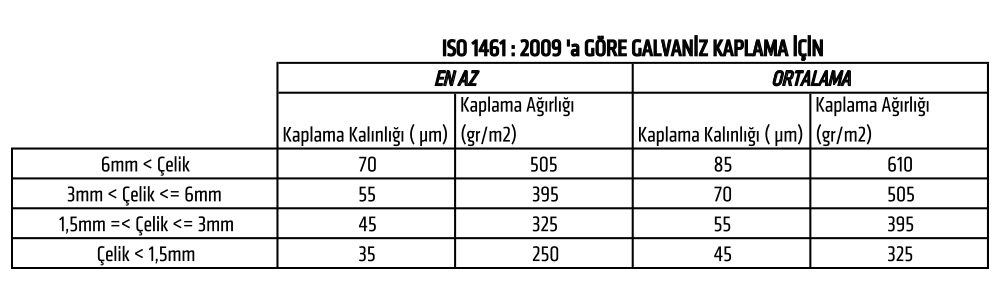
The coating thickness of galvanized products is measured using a digital micrometer. Products with coating defects are repaired or re-galvanized as necessary.
After inspection and maintenance, the products are stacked in a way suitable for loading and prepared for shipment.